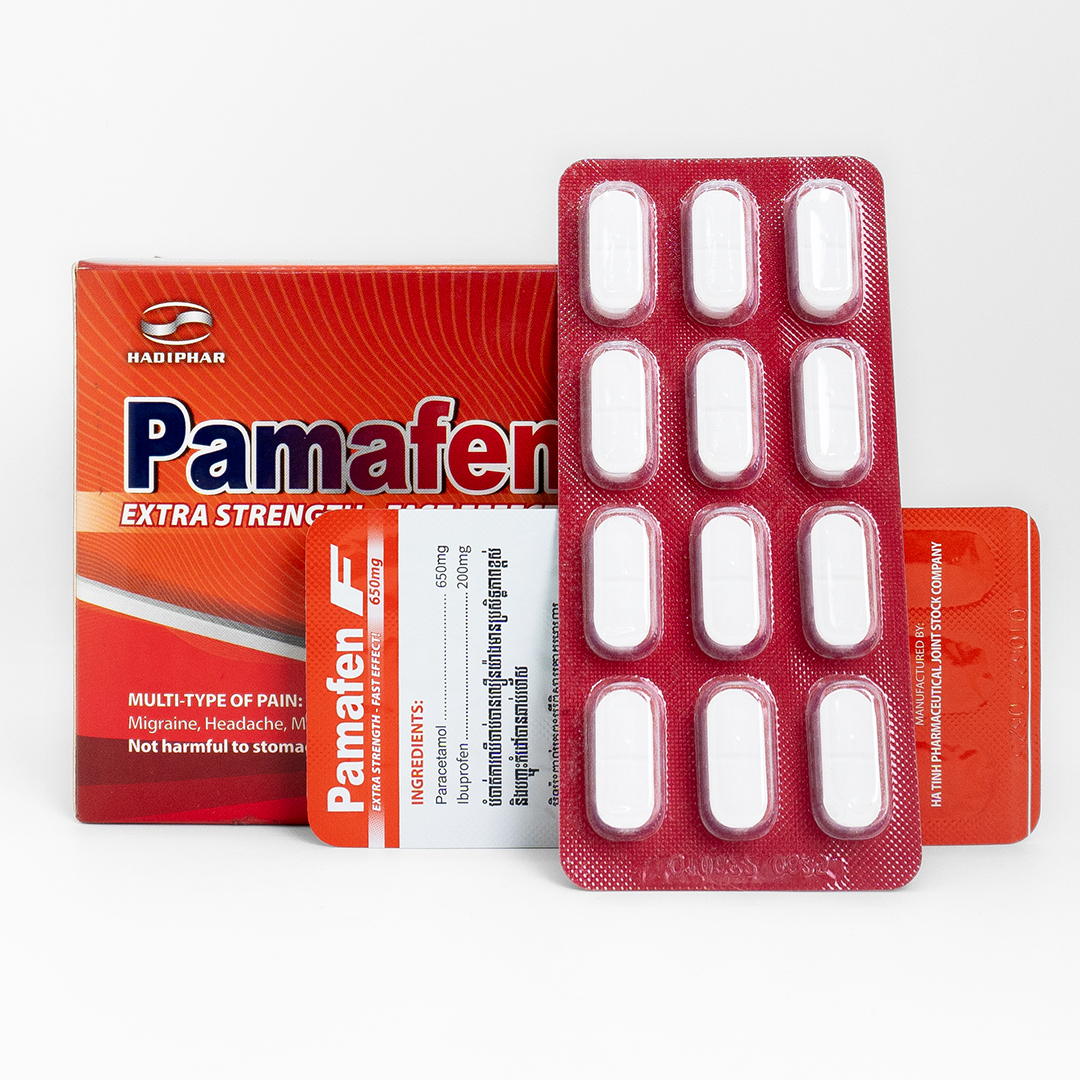
INDICATION:
Effective in treating fever
Reduce pain from: flu and cold, headache, migraine, toothache, muscle pain, backache, arthritis, stomachache.
DOSAGE AND METHOD OF ADMINISTRATION:
Usage: Taken orally with water.
Dosage:
Doses depend on body weight and age.
- Adults and adolescents weighing over 50 kg aged over 15 years: ½-1 tablet every 4 to 6 hours up to a maximum 3000 mg per 24 hours. If symptoms persist longer than 3 days, a doctor should be contacted. For continuous use only under medical supervision.
- Adolescents weighing over 40 kg aged over 12 years: The single dose ranges from 15mg/kg/day (½ tablet) to a maximum 60mg/kg/day (5x½ tablet).
PAMAFEN F are not suitable for children under 12 years and for adolescents over 12 years under 40 kg of body weight respectively, since the dose strength for this age group is not suitable. For this patient group other formulations and dose strength are available.
In patients with impaired hepatic or renal function or Gilbert’s syndrome, the dose must be reduced or the dosing interval
Impaired liver or kidney function:
In patients with severe renal insufficiency (creatinine clearance < 10 ml/min), a dosing interval of at least 8 hours must be prolonged.
CONTRAINDICATIONS:
Hypersensitivity to the active substance or to any of the excipients
WARNING AND PRECAUTION FOR USE:
Prolonged or frequent use is discouraged. Patients should be advised not to take other paracetamol containing products concurrently. Taking multiple daily doses in one administration can severely damage the liver, in such case unconsciousness does not occur. However, medical assistance should be sought immediately. Prolonged use except under medical supervision may be harmful. In children treated with 60mg/kg daily of paracetamol, the combination with another antipyretic is not justified except in the case of ineffectiveness. Caution is advised in the administration of paracetamol to patients with moderate and severe renal insufficiency, mild to moderate hepatocellular insufficiency (including Gilbert’s syndrome), severe hepatic insufficiency (Child-Pugh >9), acute hepatitis, concomitant treatment with medicinal products affecting hepatic functions, glucose-6-phosphatedehydrogenase deficiency, haemolytic anaemia, dehydration, alcohol abuse and chronic malnutrition (see section 4.2). The hazards of overdose are greater in those with non-cirrhotic alcoholic liver disease. Caution should be exercised in cases of chronic alcoholism. The daily dose should not exceed 2000 mg in such case. Alcohol should not be used during the treatment with paracetamol. Caution is advised in asthmatic patients sensitive to aspirin, because light reaction bronchospasm with paracetamol (cross-reaction) has been reported in less than 5% of the patients tested. Abrupt discontinuation of long-term use of high-dosed analgesics, taken not as directed, may cause headache, tiredness, muscular pain, nervousness and vegetative symptoms. The withdrawal symptoms subside within a few days. Patients should be advised to consult their doctor if headaches become persistent. Do not exceed the stated dose. If symptoms persist consult a doctor. Treatment with an antidote is advised if an overdose is suspected. Interference with laboratory tests: Paracetamol may affect uric acid tests by wolframato phosphoric acid, and blood sugar tests by glucose-oxidase-peroxidase.
Interaction:
Hepatotoxic substances may increase the possibility of paracetamol accumulation and overdose. The metabolization of paracetamol is increased in patients taking enzyme-inducing drugs such as rifampicin and some antiepileptics (carbamaze- pine, phenytoin, phenobarbital, primidone). Isolated reports describe unexpected hepatotoxicity in patients taking enzyme-inducing drugs and alcohol. Probenecid causes an almost 2-fold reduction in clearance of paracetamol by inhibiting its conjugation with glucuronid acid. A reduction of the paracetamol dose should be considered for concomitant treatment with probenecid. Salicylamide may prolong the elimination t1/2 of paracetamol. Metoclopramide and domperidone accelerate absorption of paracetamol. However, concurrent use need not be avoided. Cholestyramine reduces absorption of paracetamol and therefore should not be administered within an hour following paracetamol administration. Concomitant use of paracetamol (4000 mg per day for at least 4 days) with oral anticoagulants may lead to slight variations of INR values. In this case, increased monitoring of INR values should be done during the duration of the combination and after its discontinua-tion. Isoniazid: Reduction of paracetamol clearance, with possible potentiation of its action and/or toxicity, by inhibiting its metabolism in the liver. Lamotrigine: decrease in the bioavailability of lamotrigine, with possible reduction of its effect, due to possible induction of liver metabolism. Chloramphenicol: Increased plasma concentration of chloramphenicol
Using drugs for pregnancy and lactation
Pregnancy
Epidemiological data on the oral administration of therapeutic doses of paracetamol indicate no adverse reactions on pregnancy or on the health of the fetus/newborn child. Prospective data on overdose during pregnancy showed no increased risk of malformations. Reproduction studies investigating oral administration did not indicate any signs of malformation or fetotoxicity.
Paracetamol is considered to be safe in normal therapeutic doses for short-term use as a minor analgesic/antipyretic, but patients should seek the advice of their doctor regarding its use.
Lactation
Following oral administration, paracetamol is excreted into breast milk in small quantities. To date, no adverse reactions or undesirable effects are known in association with lactation. Therapeutic doses of paracetamol can be administered during breast-feeding
Effects of the drug on the ability to drive and use machine.
The drug has no effect
UNDERSIRABLE EFFECTS
- For paracetamol
Uncommon, 1/1000 <ADR <1/100
Skin: rash
Digestion: Nausea, vomiting,
Hematology: Hematopoietic disorders (neutropenia, thrombocytopenia, leukopenia), anemia.
Kidney: Kidney disease, kidney failure if tortured for a long time.
Rarely, ADR <1/100
Skin: Stevens-Johnson syndrome, toxic epidermal necrolysis, Lyell’s syndrome, acute generalized exanthematous pustulosis.
Other: Hypersensitivity reactions.
OVERDOSE AND TREATMENT:
For Paracetamol:
- Symptom:
Paracetamol toxicity can be caused by a single toxic dose, by repeated large doses of paracetamol (eg, 7.5-10 g daily, for 1-2 days), or by long-term use. Dose-dependent hepatic necrosis is the most serious acute toxic effect of overdose and can be fatal. Nausea, vomiting, and abdominal pain usually occur within 2 to 3 hours of taking a toxic dose. Methemoglobin – blood, leading to cyanosis of the skin, mucous membranes and nails is a characteristic sign of acute poisoning with p-aminophenol derivatives; Small amounts of sulfhemoglobin may also be produced. Children tend to make methemoglobin more easily than adults after taking paracetamol.
In severe poisoning, there may be initial CNS stimulation, agitation and delirium. This may be followed by central nervous system depression; stupor, hypothermia; exhausted; rapid, shallow breathing; fast, weak, irregular pulse; low blood pressure; and circulatory failure. Vascular collapse due to relative hypoxia and because of the central inhibitory effect, this effect occurs only with very large doses. Shock can occur if multiple vasodilation. Fatal choking convulsions may occur. Usually coma occurs before sudden death or several days after coma.
Clinical signs of liver damage become apparent within 2 to 4 days after ingestion of a toxic dose. Plasma aminotransferases are elevated (sometimes very high) and plasma bilirubin levels may also be elevated; Furthermore, when the liver is severely damaged, the prothrombin time is prolonged. It is possible that 10% of patients with untreated poisoning have severe liver damage; of which 10% to 20% eventually die of liver failure. Acute renal failure also occurs in some patients. Liver biopsy showed central lobular necrosis with the exception of the periportal region. In non-fatal cases, liver damage can be reversible in weeks or months.
- To solve:
Early diagnosis is important in the treatment of paracetamol overdose. There are methods for the rapid determination of drug plasma concentrations. However, treatment should not be delayed pending test results if the history suggests a serious overdose. When intoxication is severe, it is important to provide active supportive treatment. Gastric lavage in all cases, preferably within 4 hours of ingestion.
The primary detoxification therapy is the administration of sulfhydryl compounds, which presumably work in part by replenishing the liver’s stores of glutathione.
N-acetylcysteine is effective when taken orally or intravenously. The medicine must be given immediately if it has not been 36 hours since paracetamol has been taken. Treatment with N-acetylcysteine is more effective when the drug is administered less than 10 hours after taking paracetamol. When giving orally, dilute the N-acetylcysteine solution with water or a non-alco- holic beverage to get a 5% solution and must be drunk within 1 hour after mixing. Give oral N-acetylcysteine with a first dose of 140 mg/kg, then give another 17 doses, each dose of 70 mg/kg every 4 hours. Discontinue treatment if plasma paracetamol test indicates a low risk of hepatotoxicity.
Adverse effects of N-acetylcysteine include skin rash (including urticaria, which does not require discontinuation), nausea, vomiting, diarrhea, and anaphylactic reactions.
If N-acetylcysteine is not available, methionine can be used. Alternatively, activated charcoal and/or salt bleach can be used, which may reduce paracetamol absorption.
For ibuprofen:
- Symptom:
Overdose symptoms may include nausea, vomiting, stomach pain, drowsiness, black or bloody stools, coughing up blood, shallow breathing, fainting, or coma.
- To solve:
Seek emergency medical attention.
PHARMACOLOGICAL PROPERTIES:
PHARMACODYNAMICS
Paracetamol is an effective pain reliever fever reducer. Paracetamol lowers body temperature in febrile patients, but rarely lowers body temperature in normal people. The drug acts on the hypothalamus causing hypothermia, increased heat loss due to vasodilation and increased peripheral blood flow.
Ibuprofen is a non-steroidal anti-inflammatory drug with analgesic, antipyretic and anti-inflammatory effects. Ibuprofen inhibits prostaglandin synthetase and thereby prevents the production of prostaglandins, thromboxane and other products of cyclooxygenase.
PHARMACOKINETICS
Paracetamol is readily absorbed from the gastro-intestinal tract with peak plasma levels occurring about 30 minutes to 2 hours after ingestion.
It is metabolised in the liver (90-95%) and excreted in the urine mainly as the glucuronide and sulphate conjugates. Less than 5% is excreted unchanged.
The elimination half life of Paracetamol varies from about 1 to 4 hours. Plasma protein binding is negligible at usual therapeu- tic doses but increases with increasing concentrations.
A minor hydroxylated metabolite (N-acetyl-p-benzoquinoneimine) which is usually produced in very small amounts by mixed-function oxidases in the liver and which is usually detoxified by conjugation with liver glutathione may accumulate following paracetamol overdosage and cause liver damage.
The time to peak plasma concentration of paracetamol is 0.5 to 2 hours, the time to peak effect 1 to 3 hours and the duration of action 3 to 4 hours.
Ibuprofen is rapidly absorbed following administration and is rapidly distributed throughout the whole body. The excretion is rapid and complete via the kidneys.
Maximum plasma concentrations are reached 45 minutes after ingestion if taken on an empty stomach. When taken with food, peak levels are observed after 1 to 2 hours. These times may vary with different dosage forms.
MANUFACTURED BY: HA TINH PHARMACEUTICAL JOINT STOCK COMPANY
No.167 Ha Huy Tap Street, Nam Ha Ward, Ha Tinh City, Ha Tinh Province, Vietnam.