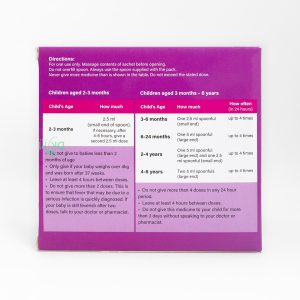
Overdose and Treatment:
For Cetirizine Hydrochloride
Symptoms:
- Observed symptoms of overdose mainly relate to central nervous system (CNS) effects, such as somnolence, dizziness, and other anticholinergic effects.
- Adverse events after consumption exceeding five times the recommended dose include: confusion, diarrhea, dizziness, fatigue, headache, malaise, mydriasis, pruritus, restlessness, sedation, somnolence, stupor, tachycardia, tremor, and urinary retention.
Treatment:
- No specific antidote is available for cetirizine.
- Supportive treatment and symptomatic management are recommended.
- Gastric lavage can be considered shortly after ingestion of the drug.
- Cetirizine is not effectively removed by hemodialysis.
Pharmacological Properties:
Pharmacodynamics:
- Cetirizine hydrochloride, as an anti-H1 receptor, demonstrates significant anti-allergic activity by inhibiting the late phase of eosinophil recruitment.
- Studies have shown it to be effective in allergic rhinitis, urticaria, and conjunctival allergies.
- Clinical trials revealed no significant QT interval prolongation when used appropriately.
Pharmacokinetics:
- Achieves peak plasma concentration within 0.5 to 1 hour.
- Food does not alter the extent of absorption but may decrease the rate of absorption.
Storage:
- Store in a cool and dry place below 30°C.
- Protect from direct sunlight and handle carefully.
Packing Box:
- 10 blisters × 10 film-coated tablets.
Shelf Life:
- 36 months from the manufacturing date.
Manufactured by:
TRUONG THO PHARMA, JSC
- Head Office: No. 93 Linh Lang St., Cong Vi Ward, Ba Dinh District, Hanoi City, Vietnam.
- Factory: Lot N3, Hoa Xa Industrial Zone, Nam Dinh Province, Vietnam.
- Tel: +84-243-7666 912 | +84-243-7666 913
Imported & Marketed by:
World Life Pharma Co., Ltd.
- S/K Chak Angre Krom, Phnom Penh, Cambodia.