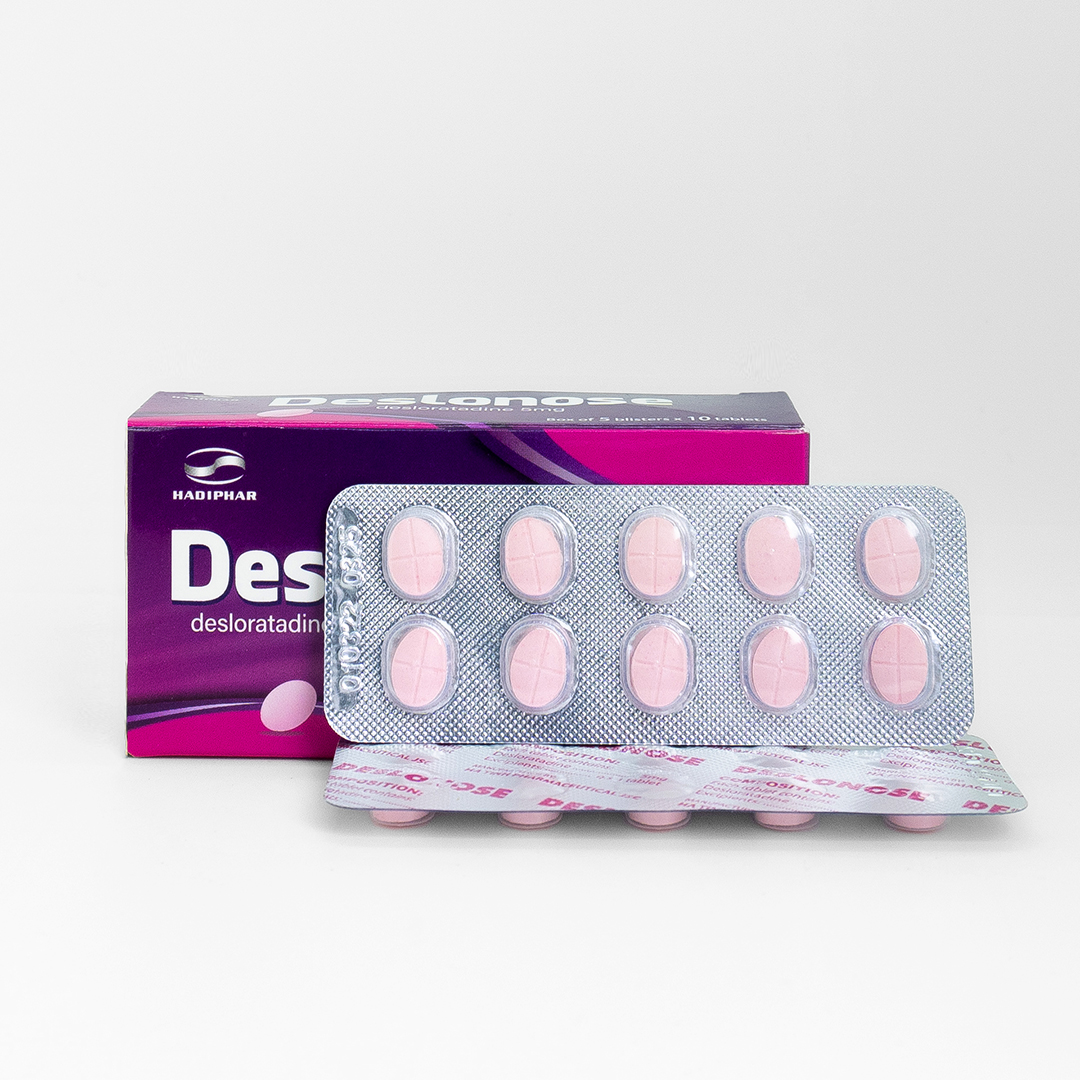
Indication:
- Allergic rhinitis
- Chronic idiopathic urticaria
Dosage and Administration:
- Adults and children ≥ 12 years old: one tablet per day, drink with or without meals.
- In patients with hepatic or renal impairment, the recommended dose of desloratadine is 5 mg only every other day.
Contraindications:
- Hypersensitivity to desloratadine, loratadine, or any of the excipients.
Warnings and precautions:
- Warnings: Avoid using this product in children younger than 2 years of age as safety and efficacy have not been established. For children between 2 and 12 years, dosage must follow specific medical recommendations after proper assessment of the appropriate weight and medical history.
- Use with caution in individuals with severe liver or kidney impairments.
Drug interactions:
- Avoid using Desloratadine concurrently with alcohol, anticholinergic drugs, or medications affecting liver enzyme metabolism.
Use during pregnancy and breastfeeding:
- Pregnant women: Use only if absolutely necessary under strict medical guidance.
- Breastfeeding women: Desloratadine is excreted into breast milk, so consult your physician before use.
Effects on ability to drive and use machines:
- Patients should exercise caution as dizziness or drowsiness may occur in some cases.
Overdose and treatment:
- Symptoms of overdose may include drowsiness, dizziness, and dry mouth. Seek immediate medical attention.
Desloratadine reduces the concentration and effect of acetylcholinesterase inhibitors, benzylpenicillin glycosides, bethanechol, ambenonium, physostigmine.
Food does not affect the bioavailability of desloratadine.
Patients should avoid alcohol because desloratadine interacts with drugs affecting hepatic metabolism, including alcohol and other sedative agents (benzodiazepines, antihistamines).
Undesirable effects:
Rare cases of adverse events were noted.
Overdose and treatment:
In clinical studies, overdose was detected when desloratadine was used up to 45 mg/day (9 times the therapeutic dose). In case of overdose, treat as usual, including reduction of absorption and symptomatic treatment. Desloratadine can be eliminated by peritoneal dialysis or hemodialysis.
Pharmacokinetics:
Desloratadine is the main active metabolite of loratadine. It is used once per day in symptomatic allergic conditions such as rhinitis or chronic urticaria. Desloratadine has high bioavailability when taken orally, with a half-life elimination of about 27 hours. Excretion occurs primarily through hepatic metabolism.
Pharmacodynamics:
Desloratadine does not inhibit the central nervous system, dries the 5-HT serotonin receptors, which leads to less antihistaminic effect.
Pack sizes:
Pack sizes: 20 tablets
Shelf life: 36 months since manufacture date.
Note: Keep out of reach of children. Read instructions carefully before use.
MANUFACTURED BY: HA TINH PHARMACEUTICAL JSC
No.167 Ha Huy Tap Street, Nam Ha Ward, Ha Tinh City, Ha Tinh Province, Vietnam.