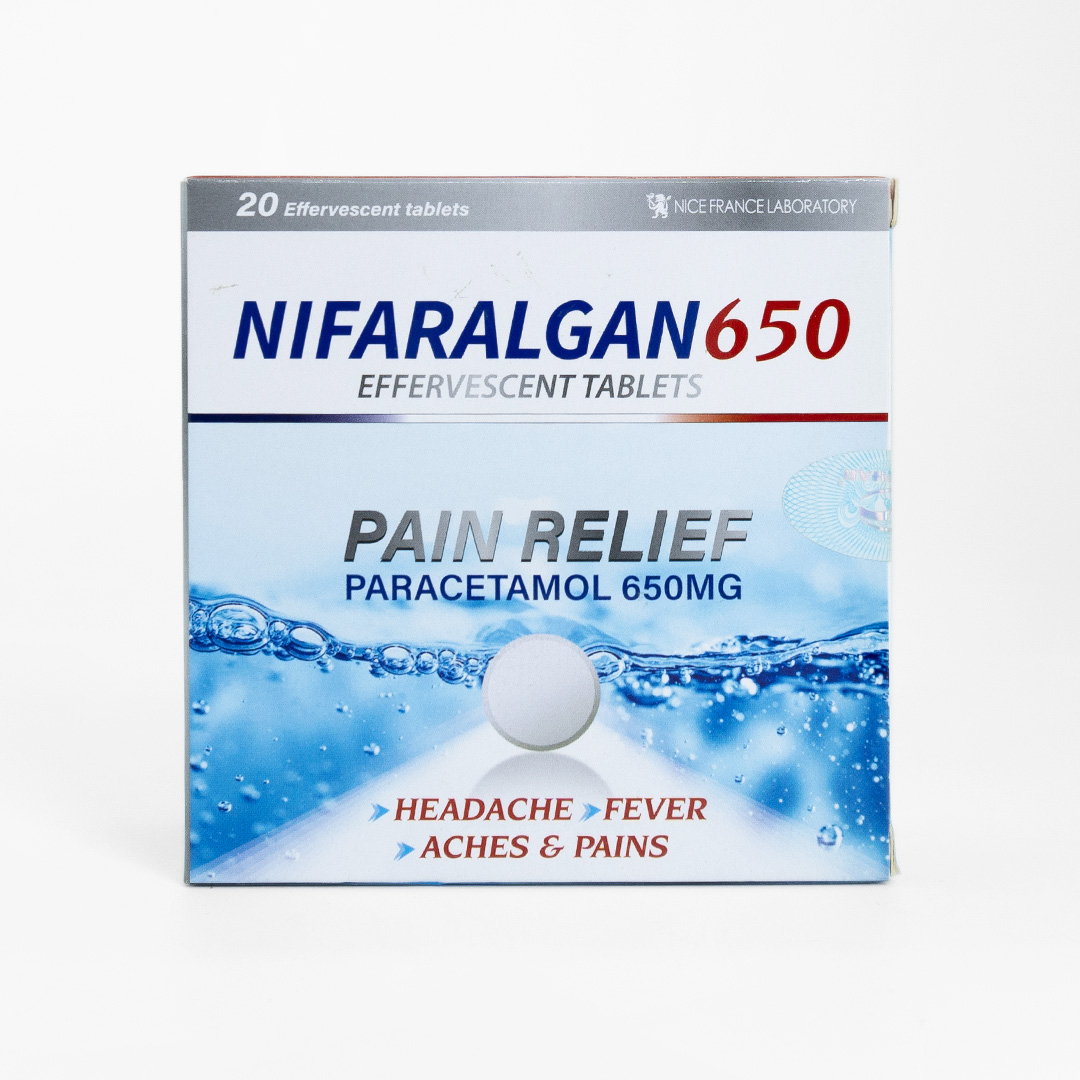
POSOLOGY & ADMINISTRATION:
Posology
- Adults including the elderly and children over 16 years: One tablet every 4–6 hours as required, to a maximum of 6 tablets daily in divided doses.
- Children 10–15 years: 1/2–1 tablet (based on weight of the children) every 4–6 hours as necessary to a maximum of 4 doses in 24 hours.
- Children under 10 years: Not recommended for children under 10 years of age. Alternative presentations of paracetamol are recommended for pediatric usage in order to obtain suitable doses of less than 650mg.
- Recommended dosage of paracetamol is 10–15 mg/kilogram of body weight. Interval between each dosage is at least 4–6 hours. The patient should consult a physician or calculate the suitable dosage to use safely and effectively.
Method of Administration
For oral administration. The tablets should be placed in a full tumbler of water immediately before use and allowed to dissolve completely before swallowing.
CONTRAINDICATION:
- Patients hypersensitive to ingredients of the drug.
- Patients with severe liver failure, glaucoma, urinary retention due to urethral disorder, prostate, anemia, or have heart disease, lung, kidney.
- Patients with glucose-6-phosphate dehydrogenase deficiency disease.
- Breastfeeding, infants, and premature babies.
INTERACTION:
- Intake of paracetamol in long time and high dose slightly increase the anticoagulant effect of coumarin and indandion derivates. Use product together with phenothiazine and heat-reduction therapy can lead to serious over heat-reduction consequence.
- Anticonvulsants (phenytoin, barbiturate, carbamazepine), isoniazid, and anti-tuberculin drugs may increase the toxicity to the paracetamol to liver.
- Excessive and long-term drinking may increase the risk of paracetamol poisoning the liver. Cholestyramine reduces paracetamol absorption (not drinking within 1 hour).
ADVERSE DRUG REACTION:
Common, 1/1000 < ADR < 1/100
-
- Skin: Skin rash.
- Gastrointestinal: Nausea, vomiting.
- Hematology: Hematopoietic disorders (neutropenia, reduction of whole blood cells, neutropenia), anemia.
- Renal: Kidney disease, kidney toxicity in long-term abuse
Rarely, ADR < 1/1000
-
-
- Skin: Stevens-Johnson syndrome, toxic epidermal necrolysis, Lyell’s syndrome, acute onset epiglottis.
- Other: Hypersensitivity reaction.
-
Caution:
Children and adults with undesirable side effects are more likely to be associated with cholinergic resistance and back stimulation (e.g., restlessness, anxiety). Inform your doctor if any of the side effects occur when using the medicine.
PRECAUTION:
Paracetamol is relatively non-toxic with therapeutic doses. However, to urinary phenylecton patient or person who have to limit the amount of phenylalanine intake, should avoid use paracetamol with drug or food containing aspartame. Caution in use at patient having prehistory of anemia, liver and kidney failure. Intake of alcohol drinks or wine can increase the toxic of paracetamol to liver, therefore patient should avoid alcohol and wine during use of product.
PHARMACOLOGICAL PROPERTIES:
Pharmacodynamic
Paracetamol: Paracetamol is thought to act primarily in the CNS, increasing the pain threshold by inhibiting both isoforms of cyclooxygenase, COX-1, COX-2, and COX-3 enzymes involved in prostaglandin (PG) synthesis. Unlike NSAIDs, Paracetamol does not inhibit cyclooxygenase in peripheral tissues and, thus, has no peripheral anti-inflammatory effects. While aspirin acts as an irreversible inhibitor of COX and directly blocks the enzyme’s active site, studies have found that Paracetamol indirectly blocks COX, and that this blockade is ineffective in the presence of peroxides. This might explain why Paracetamol is effective in the central nervous system and in endothelial cells but not in platelets and immune cells which have high levels of peroxides.
Studies also report data suggesting that Paracetamol selectively blocks a variant of the COX enzyme that is different from the known variants COX-1 and COX-2. This enzyme is now referred to as COX-3. Its exact mechanism of action is still poorly understood, but future research may provide further insight into how it works. The antipyretic properties of Paracetamol are likely due to direct effects on the heat-regulating centers of the hypothalamus resulting in peripheral vasodilation, sweating, and hence heat dissipation.
Pharmacokinetic
Paracetamol: Paracetamol is well absorbed in the gastrointestinal tract. Oral bioavailability is dose dependent; with larger doses, the hepatic first-pass effect is reduced due to overwhelming of the liver enzymatic capacity; and therefore, bioavailability is increased. Rectal administration of paracetamol is also feasible. In this case, bioavailability is inconsistent and in overall reduced, due to incomplete dissolution of the suppository in the rectum. The absorption rate through this route of administration is elongated.
Paracetamol is distributed throughout the body fluids in a homogeneous way. The analgesic activity is attributable to the small fraction that penetrates into the brain. Paracetamol given at therapeutic doses binds to plasma proteins at less than 20%. In case of intoxication, this proportion may increase to up to 50%.
Paracetamol is essentially metabolized in the liver by conjugation with glucuronic acid (55%) and sulfuric acid (35%). Hepatotoxic metabolites are produced in small amounts by the cytochrome P450 (isoenzyme CYP2E1). In the therapeutic plasma concentration range, this metabolite is detoxified by conjugation with glutathione. In case of intoxication the amount of this toxic metabolite increases and outweighs the amount of available glutathione, which can lead to hepatic failure and renal tubular necrosis.
Metabolites are excreted through the kidneys in the urine. Only 2–5% of the dose is excreted in an unchanged form in the urine. As a consequence of its short elimination half-life (1–3h), 24 hours after the ingestion of a single dose of paracetamol, 98% of the dose is eliminated.