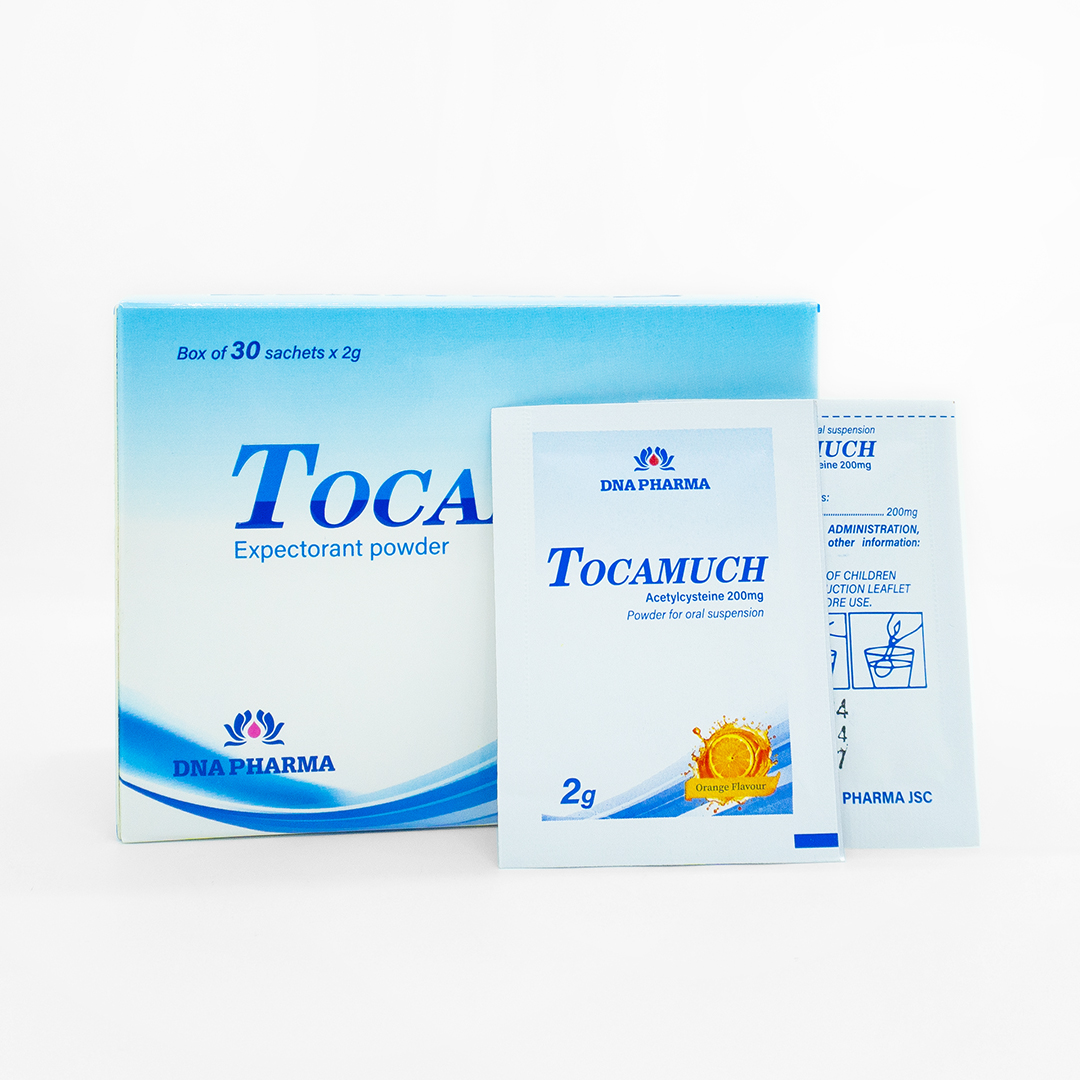
INDICATION: Mucolytic adjuvant in the therapy of mucoviscidosis, respiratory disorders associated with thick, viscous mucus hypersecretion, for example acute and chronic bronchitis, routine cleaning of tracheostomy. Use as antidote in paracetamol overdose.
CONTRAINDICATION: Prehistory of asthma or risk of bronchospasm with any medicine containing Acetylcysteine. Hypersensitive with Acetylcysteine.
THERAPEUTIC INDICATION:
Mucolytic adjuvant in the therapy of mucoviscidosis, respiratory disorders associated with thick, viscous, mucus hypersecretion, for example acute and chronic bronchitis, routine cleaning of tracheostomy.
Use as antidote in paracetamol overdose.
POSOLOGY AND METHOD OF ADMINISTRATION:
For Mucolytic adjuvant:
- Kids from 2 to 6 years old: 1 sachet/time x 2 times/day.
- Adult and kids above 6 years old: 1 sachet/time x 3 times/day.
INTERACTION:
- Antitussive drugs and acetylcysteine should not be administered concomitantly because reducing the cough reflex may lead to a build-up of bronchial secretions.
- Activated charcoal may reduce the effect of acetylcysteine.
- It is advisable not to mix Acetylcysteine 200 mg Powder for Oral Solution with other medicinal products.
- In vitro tests have shown that when cephalosporin antibiotics and acetylcysteine are mixed, there is a degree of antibiotic inactivation. It is precautionary to advise the administration of oral antibiotics at least two hours before or after acetylcysteine.
- Concurrent administration of nitroglycerin and acetylcysteine causes significant hypotension and leads to temporal artery dilation with possible onset of headache.
- If concurrent administration of nitroglycerin and acetylcysteine is required, patients should be monitored and warned for hypotension that can be severe and accompanied by a headache.
The occurrence of serious skin reactions such as Stevens-Johnson syndrome and toxic epidermal necrolysis have been reported in temporal association with the use of acetylcysteine. In most of these cases reported at least one other drug was administered at the same time, which may have possibly enhanced the described mucocutaneous effects.
In case of recurrence skin and mucosal lesions, medical advice should be sought at once and the use of acetylcysteine terminated immediately.
PRECAUTION:
- Patients with bronchial asthma should be closely monitored during therapy; if bronchospasm occurs, treatment with TOCAMUCH should be discontinued immediately.
- Administration of acetylcysteine, especially at the beginning of treatment, may liquefy bronchial secretions and, at the same time, increase their volume. If the patient is unable to expectorate efficiently, to avoid retention of secretions postural drainage and tracheal suction should be used.
- There are no studies on the efficacy and safety of acetylcysteine 200 mg three times daily in adolescent population. However, mild to severe adverse reactions have been reported with the use of acetylcysteine in adults and adolescents.
- This medicine contains aspartame, which is a source phenylalanine. This may be harmful to people with phenylketonuria.
- Acetylcysteine can cause interference with the colorimetric assay method for the determination of salicylates.
- Acetylcysteine can interfere with tests for ketones in urine.
BREASTFEEDING AND PREGNANCY:
- Pregnancy: Animal studies do not indicate direct or indirect harmful effects with respect to reproductive toxicity. As a precautionary measure, it is preferable to avoid the use of TOCAMUCH during pregnancy.
- Breastfeeding: There is insufficient information on the excretion of acetylcysteine in human milk. A risk to the newborns/infants cannot be excluded.
DRIVING AND OPERATING MACHINE:
No studies on the effects on the ability to drive or use machines have been performed. TOCAMUCH has no known effect on the ability to drive and use machines